Published Ryan Schonert on December 2, 2019
Nitrogen is the perfect replacement carrier gas for those who want to lower helium usage but can’t run hydrogen. Like helium, nitrogen isn’t flammable, so it’s safe, and unlike helium, it’s quite cheap. As I mentioned in Part 1, the longer runtimes required to achieve proper separation are arguably the biggest reason for avoiding nitrogen. Not only do longer run times mean a longer wait before samples are finished, you also take a hit to your sensitivity, as your peaks will become broader and shorter. If you speed up the run using something like a speed-optimized translation, runtime and sensitivity do improve, but you lose resolution and risk getting bad data. Neither of these scenarios are ideal, but we think that GC-VUV can be the solution to these problems.
To see how GC-VUV performed using other carrier gases, we analyzed various gasolines using helium, hydrogen, and nitrogen. If you haven’t already, go check out Parts 1 and 2 for the experimental details and the results for hydrogen. Otherwise, let’s take a look at nitrogen. Figure 1 shows the relevant conditions that we used for helium and nitrogen.

Figure 1. The relevant conditions altered for each method. The flow rate and oven programming were determined using a method translator. The acquisition frequency was determined by taking the number of scans in a traditional D8071 run and calculating a new frequency to give the same approximate number of scans in the new runtime.
Just as we did with hydrogen, we ran a pure translation, a speed optimized translation, and a method with a forced runtime of 33.6 minutes. For hydrogen, the forced 33.6-minute runtime was actually slower than the other two methods, so we saw an increase in resolution; however, with nitrogen, the 33.6-minute runtime is compressing the chromatography even further, decreasing resolution. Other detectors might not have been able to handle the resulting coelutions, but we believed that VUV could really shine here. How did we do? Let’s see!
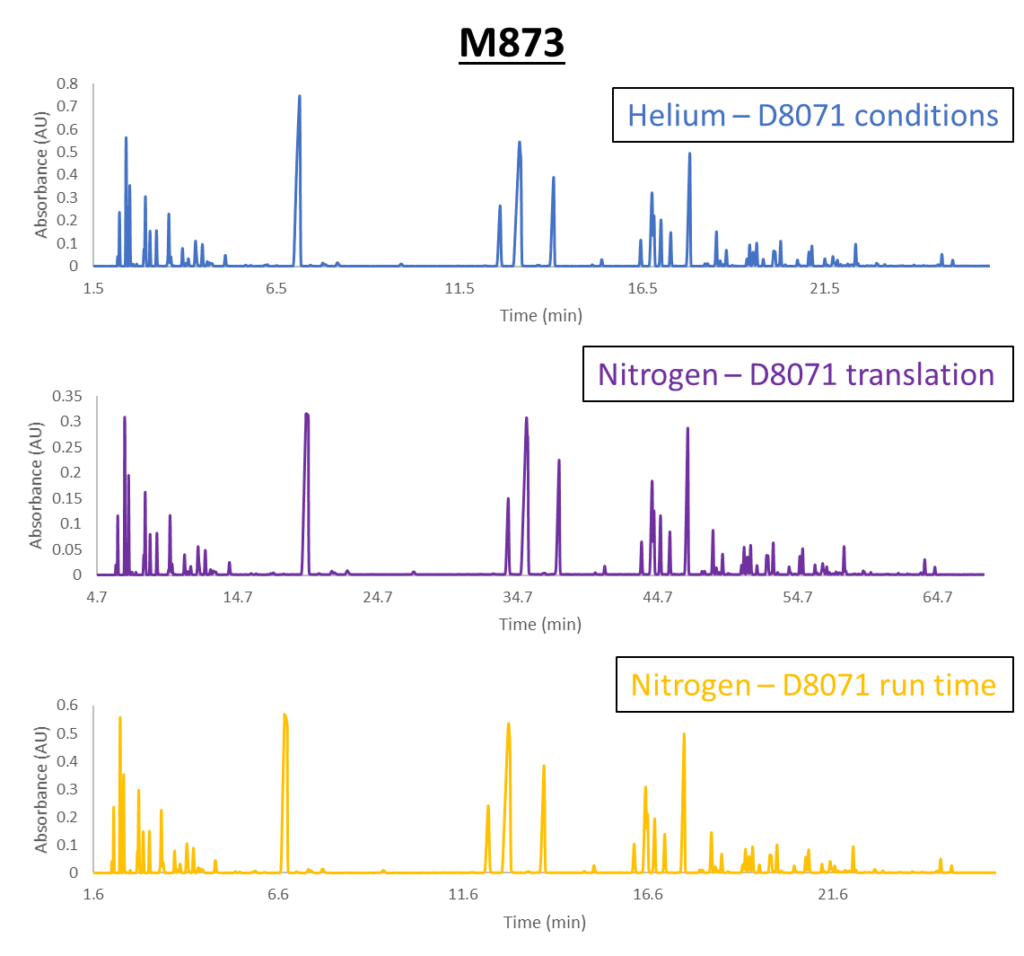
Figure 2. Comparison of the M873 gasoline sample run under ASTM D8071 conditions with helium (top), translated D8071 conditions with nitrogen (middle), and a forced runtime of 33.6 minutes with nitrogen (bottom). The peaks in each chromatogram line up very well despite the differences in method conditions, which means that the data for each run should be similar as well.
We can take a look at Figure 2 and see the power of method translation at work. Like we did with hydrogen, we’re able to get nearly equivalent peak separation on helium and nitrogen, plus the peak separation is still pretty good even when we compress the nitrogen run to 33.6 minutes. If we compare each of the three nitrogen methods, we can see slight decreases in peak resolution every time the run time is shortened, as shown below in Figure 3. However, using VUV’s spectral deconvolution, we can buy back that lost resolution, allowing us to obtain good data each time.
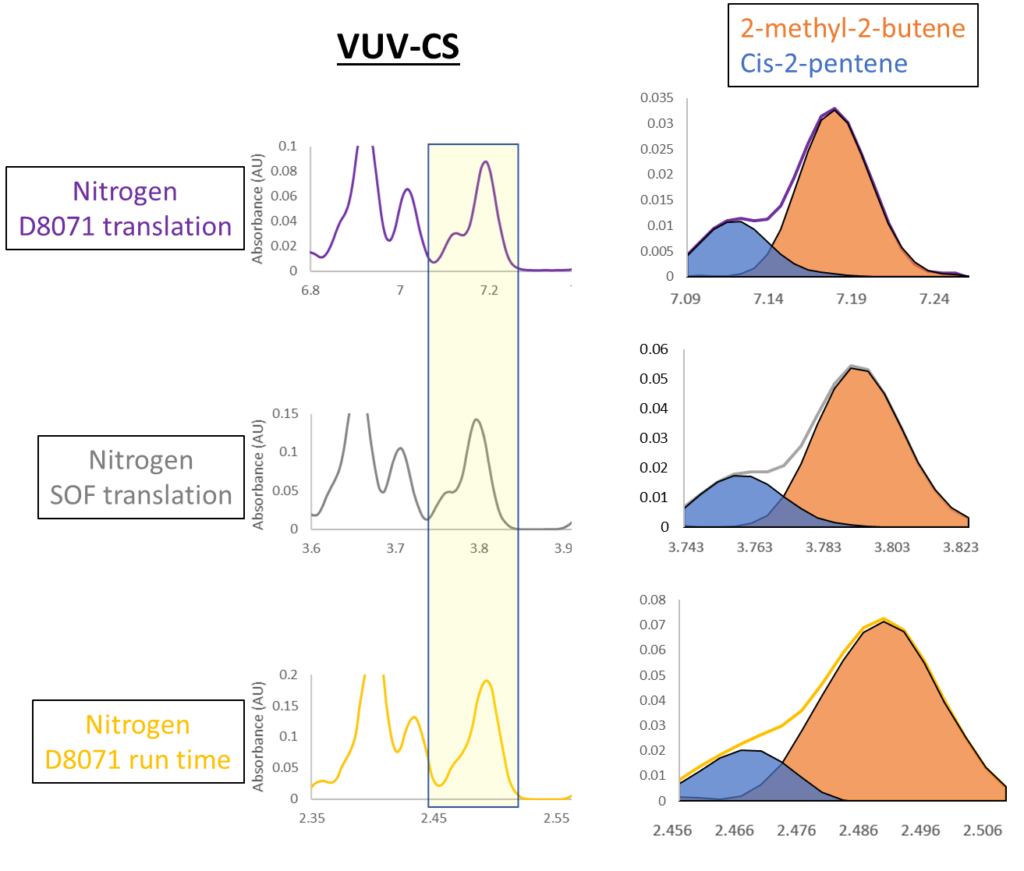
Figure 3. The coelution of 2-methyl-2-butene and cis-2-pentene in each of the nitrogen runs for the VUV-CS gasoline sample. The total run time of each run decreases from top to bottom, causing a gradual decrease in resolution, but VUV is still able to pull these compounds apart and quantify them individually. Notice that while resolution decreases from top to bottom, the scale on the Y axis indicates increasing sensitivity as run time decreases.
Table 1 shows the PIONA data for this sample. Like before, we see very comparable numbers between each of the gasoline runs.
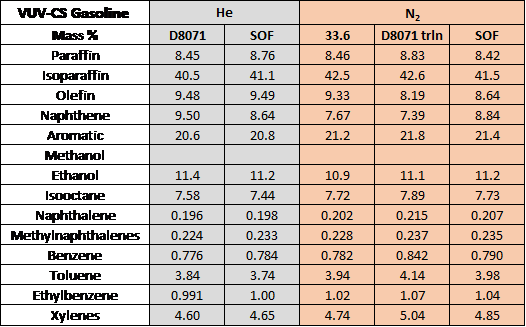
Table 1. Data gathered for VUV-CS gasoline using both with helium and nitrogen. D8071 – original ASTM D8071 conditions; SOF – speed-optimized flow conditions; D8071 trln – pure translation of ASTM D8071 conditions to hydrogen; 33.6 – forced runtime of 33.6 minutes to match the runtime of ASTM D8071. Although there are some slight differences between each of the runs, the data is overall very comparable, meaning that nitrogen can be used effectively as a replacement for helium. VUV’s spectral deconvolution is a powerful tool, but it really comes in handy in this case. Switching between carrier gases and changing the run conditions can have impacts on chromatography, and these impacts could be serious issues if you’re using a different detector, but GC-VUV brings a lot of flexibility to the table. It’s not a perfect solution, as we do see slight differences in the data between the runs for each gasoline sample. If you are curious about how each of the 6 samples fared under both hydrogen and nitrogen conditions, please feel free to reach out so we can discuss the data! We would love to hear your feedback.
As I mentioned in Part 1, switching carrier gases comes with some caveats to consider. Thankfully, unlike hydrogen, nitrogen is very safe and can be handled easily. It’s also very cheap relative to helium. As discussed previously, run times obtained using nitrogen are very long compared to hydrogen or helium. Thankfully, VUV allows us to shorten our run times with spectral deconvolution. Other detectors may have various issues with nitrogen; for example, nitrogen isn’t great for mass spectrometry. Nitrogen molecules tend to scatter sample ions more than helium, and the increased ionization of nitrogen molecules may decrease the number of electrons available to ionize sample compounds, so nitrogen can cause a hit to MS sensitivity. Again, VUV shines in this area, as the VGA-100 and VGA-101 are fully compatible with nitrogen carrier gas.
We believe that both nitrogen and hydrogen give great approximations of data gathered using helium, and both could serve as a viable option for reducing your lab’s helium usage. As the world continues to use up helium and its prices go up, the scientific community needs to be ready. Thankfully, GC-VUV is one option that can make the carrier gas transition a little smoother.








Leave a Reply