It’s likely that food company giants like General Mills, Hershey’s, Kellogg’s, and Nestlé will be looking for new analytical alternatives that can rapidly identify the compounds in their vanilla sources to ensure the integrity of their products. The fact that less than 1% of vanilla flavor comes from actual vanilla orchids may also suggest that monitoring for adulteration will take on added importance in the future. I believe that the use of GC-VUV provides new analytical flavor to an industry that was a little too “vanilla” in the past.
Published Paul Johnson, VUV Analytics on September 21, 2016

It’s always interesting when market forces, industry needs, and scientific research coincidentally converge. I happened to be reading this article about the worldwide vanilla shortage in Chemical and Engineering News just as the first vanilla data generated by GC-VUV was shared with me. The lab of Dr. Kevin Schug at University of Texas at Arlington has been in the process of developing a rapid GC method for separating natural and synthetic vanilla components.
Once Nestlé vowed to eliminate artificial additives from chocolate candy sold in the U.S., it put enormous pressure on other food and flavor companies to find natural sources of vanilla which is already in short supply. A quick screening tool like GC-VUV could be very useful to food manufacturers who are interested in authenticating the sources of their vanilla. Regulators monitoring for vanilla adulteration may also find GC-VUV to be a more robust and accessible analytical solution than what exists today.
This post in LC/GC magazine shows an example of how complex vanilla characterization can be using legacy analytical techniques. It describes how HPLC is combined with headspace solid-phase microextraction (HS–SPME) and gas chromatography with mass spectrometry (GC-MS) to measure volatile, semi-volatile, and non-volatile vanilla variants. However, it states that the bulk of compounds responsible for vanilla flavor are volatile and require the use of GC-MS. This seems like a lot of time spent acquiring (GC-MS runtime in Figure 1 was >120 min.) and interpreting data.
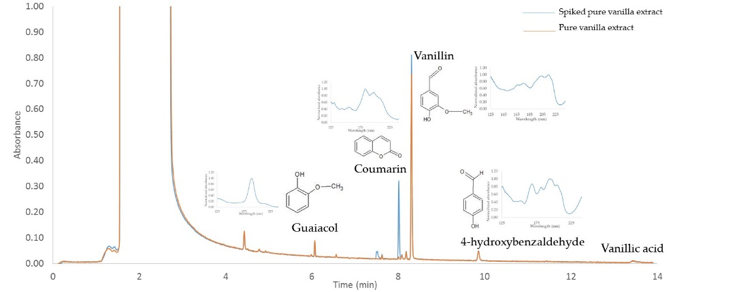
Data provided courtesy of Inês C. Santos & Kevin Schug Department of Chemistry & Biochemistry, University of Texas at Arlington
In contrast, the GC-VUV method demonstrated in the figure below was completed in less than 14 minutes. Furthermore, the identification of naturally occurring components and adulterants is unambiguous because each of these compounds have distinct spectral fingerprints that can be matched with data in the VUV library. Our GC-VUV example compares the chromatograms of natural vanilla extract and natural vanilla extract spiked with tonka bean extract. The spiked sample shows a strong presence of an unnatural compound thought to be the hepatotoxin (or in layman terms, a toxic chemical substance that damages the liver) coumarin. The VUV library verifies the identity of coumarin, and simple spectral quantitation provides its relative % mass.









Leave a Reply