Published Jack Cochran on August 16, 2018
Quantitative VUV Spectral Deconvolution of Coeluting Methylstyrene Isomers
In a previous post, I demonstrated how styrene and some alkyl substituted analogs have unique VUV absorbance spectra. I also suggested that because of their VUV spectral uniqueness, they could be deconvoluted when they coelute in gas chromatography (GC), which is something that can’t be done with GC and mass spectrometry given that their electron ionization mass spectra are essentially identical. Spectral deconvolution can be a “who cares” exercise if compounds are coeluted unnecessarily, but with methylstyrenes, GC coelutions occur naturally.
Figure 1 is an overlay of 2-, 3-, and 4-methylstyrene VUV absorbance spectra. Hopefully you can see that each spectrum is unique. The methystyrenes coelute when using a 100% dimethylpolysiloxane GC column, so quantitative determinations for these positional isomers depend on successful deconvolution, which is accomplished across relatively wide concentration ranges when using VUV spectroscopy (Figure 2 and Table 1 and Figure 3).
If isomer analysis is important for you, take the pressure off your GC system and consider a VUV spectrometer as your detector.
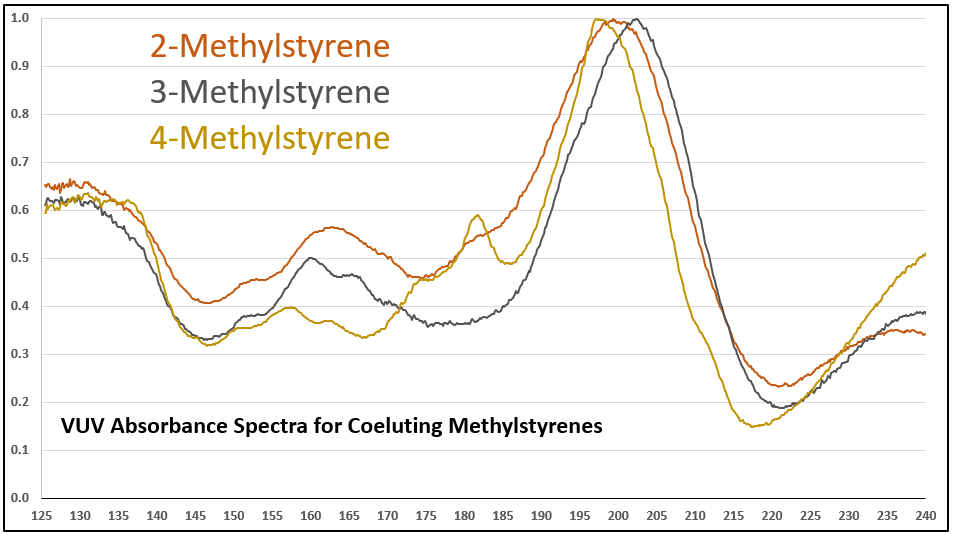
Figure 1. Overlaid VUV absorbance spectra for 2-, 3-, and 4-methylstyrenes. Each VUV spectrum is unique, unlike their mass spectra.
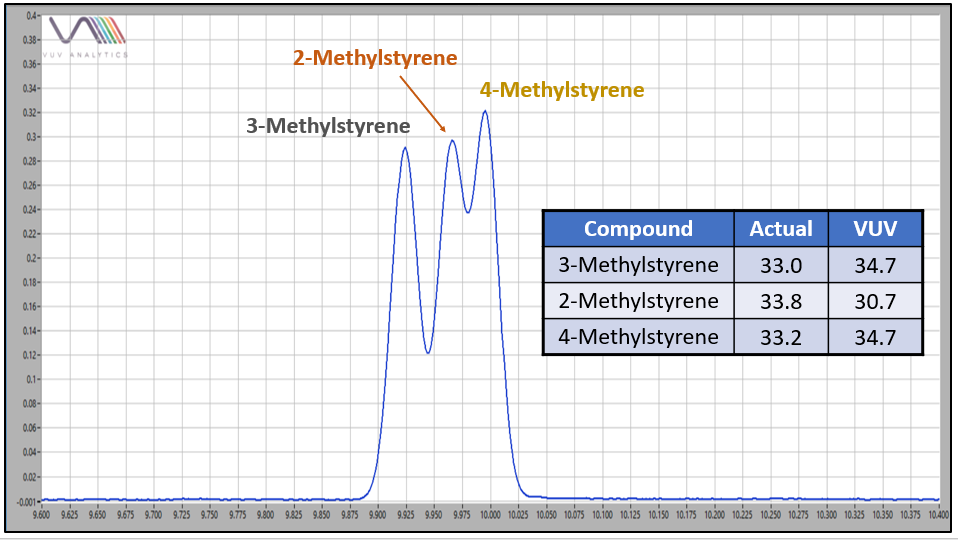
Figure 2. 2-, 3-, and 4-methylstyrenes often coelute in GC analysis. Spectral deconvolution using VUV offers the capability for quantitative estimates of each isomer. Values are in mass %.
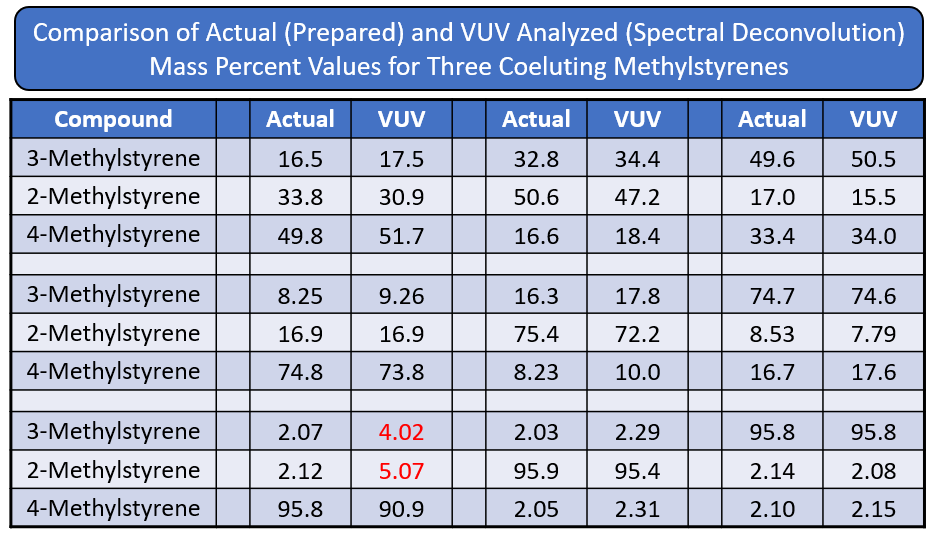
Table 1. VUV absorbance spectra offer relatively robust quantitative deconvolutions for methylstyrene positional isomers when they coelute during GC analysis, even when the concentration differentials are quite large.
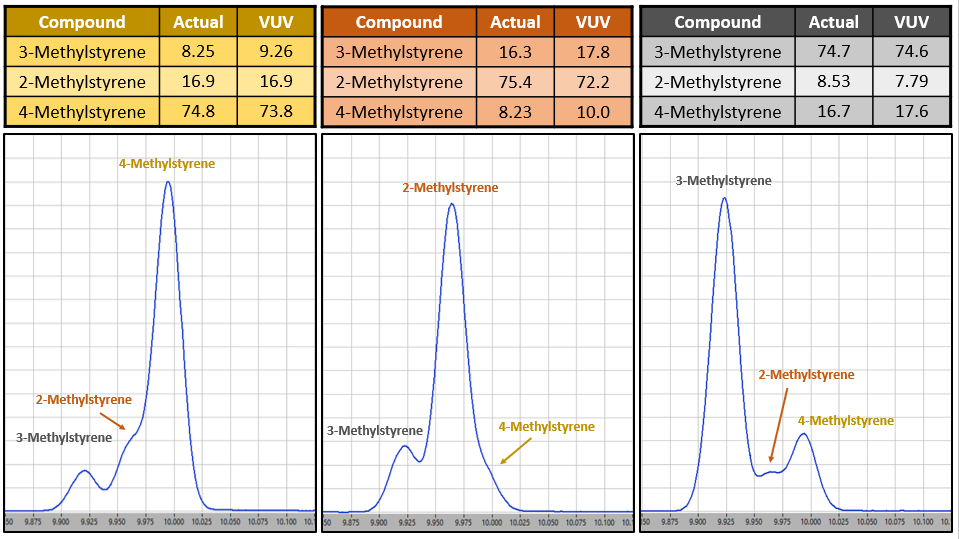
Figure 3. Methylstyrenes coelute in GC analysis, but are accurately deconvolved with VUV spectroscopy, even when concentration differentials are substantial. Values are in mass %.









Have you thought of publishing this?
Some of the figures may not impress. However, when you look at the related chromatograms you realize that detector like FID and MS cannot measure them quantitively at all!!