Published Paul Johnson, VUV Analytics on December 5, 2016
The abuse of synthetic cannabinoids by teenagers, young adults, and the homeless population has led to thousands of hospitalizations and overdose deaths over the past several years. These compounds are sold by international manufacturers and imported legally into the U.S. due to the synthesis of banned substance variants outpacing the DEA’s ability to identify them. We recently learned that the synthetic cannabinoid problem extends into to the business of legal cannabis production. A number of growers in the Pacific Northwest have been caught spraying their harvests with these synthetic compounds to increase the perceived potency of their products. Consumer safety is at risk while states like Washington, Oregon, and others work towards consensus on testing requirements and standardization of methods.
Our latest blog described how GC-VUV was used to quickly identify the structural isomers of phenylethylamine and cathinone drugs of abuse. Likewise, VUV spectroscopy is able to distinguish between the isoforms of natural and synthetic cannabinoids. All compounds absorb strongly in the VUV spectrum, and their inherent absorbance cross sections lead to unique spectral fingerprints.
Figure 1 shows a spectral overlay of the naturally occurring cannabinoids cannabinol, cannabidiol, and (-)-Δ9-tetrahydrocannabinol. The spectra are distinct despite the structural similarities between the related compounds. In Figure 2 we have compared cannabinol with the spectra of synthetic cannabinoids AM2201 and 5-fluoro-cumyl-pinaca. Cannabinol, 5-fluoro-cumyl-pinaca, and AM2201 represent the phytocannabinoid, indazole, and naphthoylindole chemical classes, respectively. It is clear that each compound class has distinct spectral characteristics, yet they share features common to all cannabinoids. VUV spectroscopy excels at not only speciating individual compounds, but also in identifying and quantitating by class. This capability was first demonstrated by the VUV PIONA+ application, but has utility in markets beyond oil and gas. Figure 3 demonstrates this point by highlighting the spectral features that are inherent to the phytocannabinoid and indazole classes.
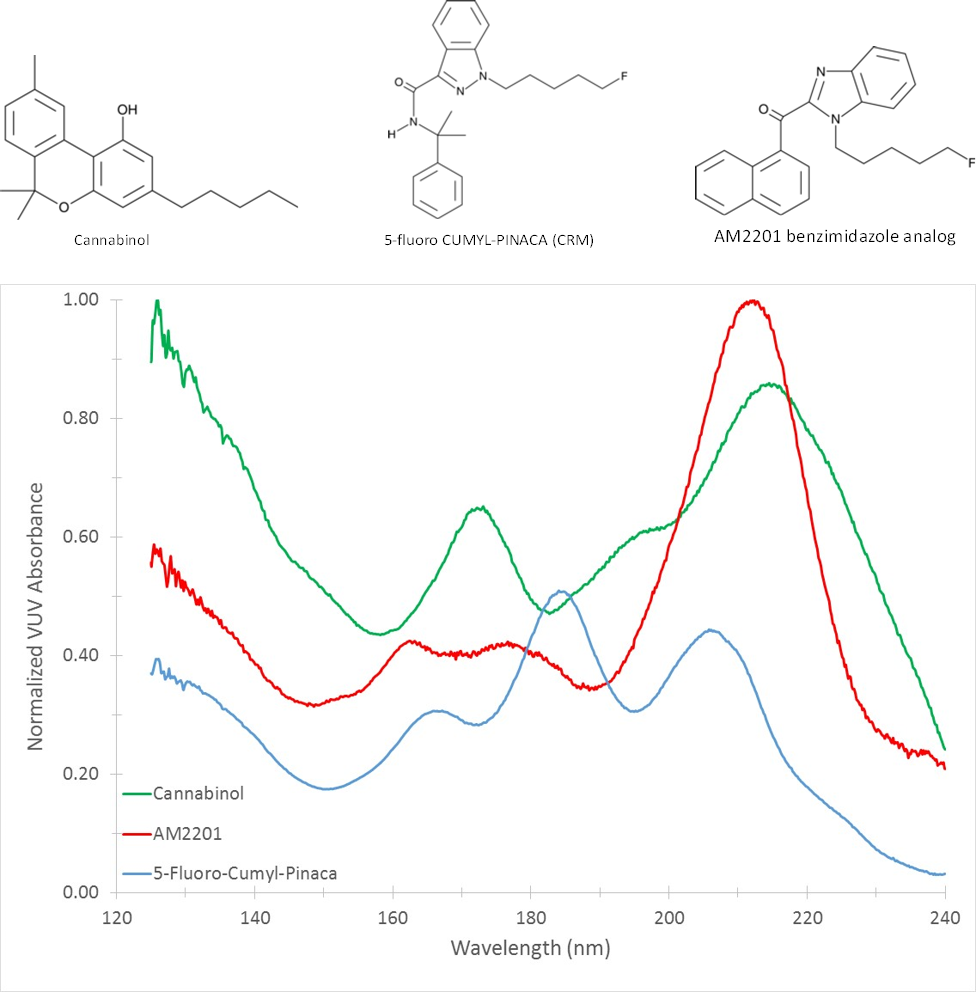
Figure 1: VUV spectra of the naturally occurring cannabinoids cannabinol, cannabidiol, and (-)-Δ9-tetrahydrocannabinol.
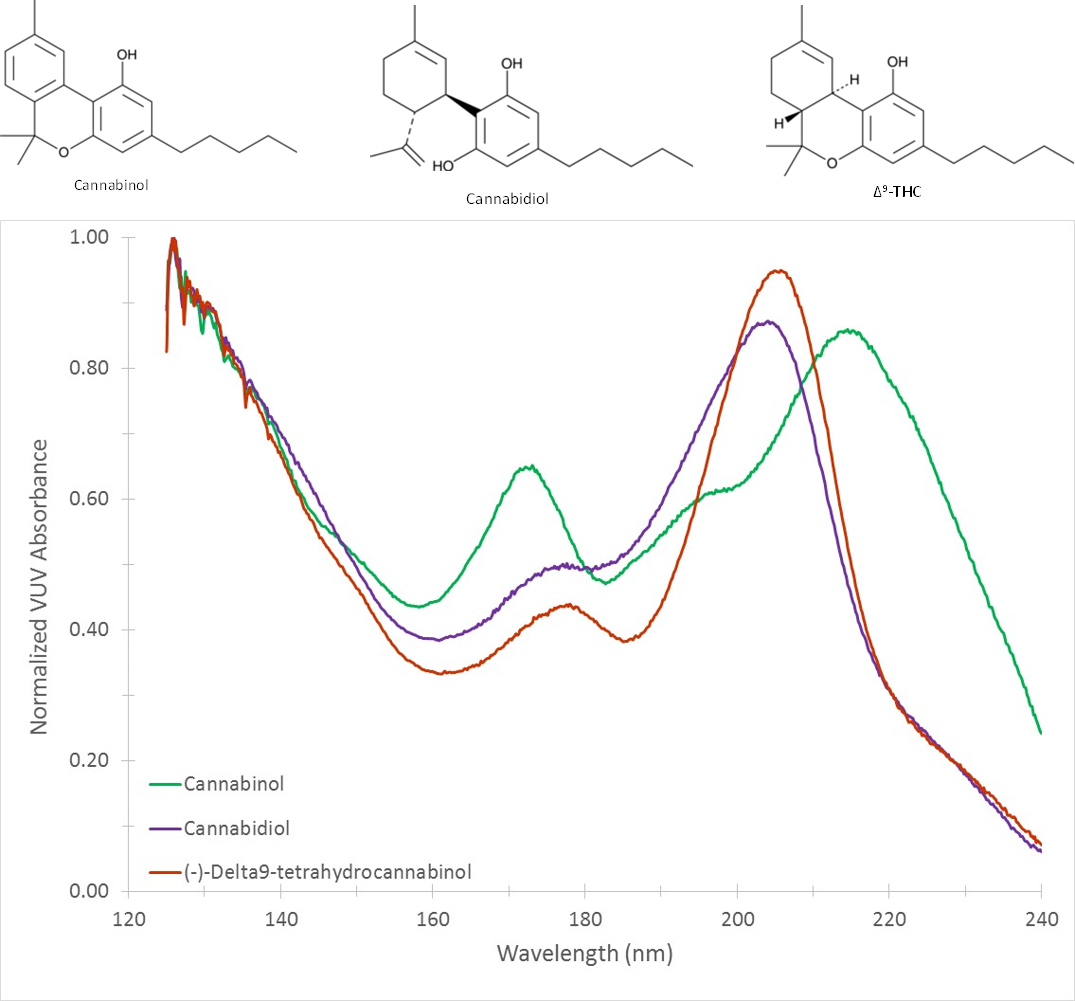
Figure 2: A VUV spectral overlay comparison of cannabinol, 5-fluoro-cumyl-pinaca, and AM2201 which represent the
phytocannabinoid, indazole, and naphthoylindole compound classes, respectively.
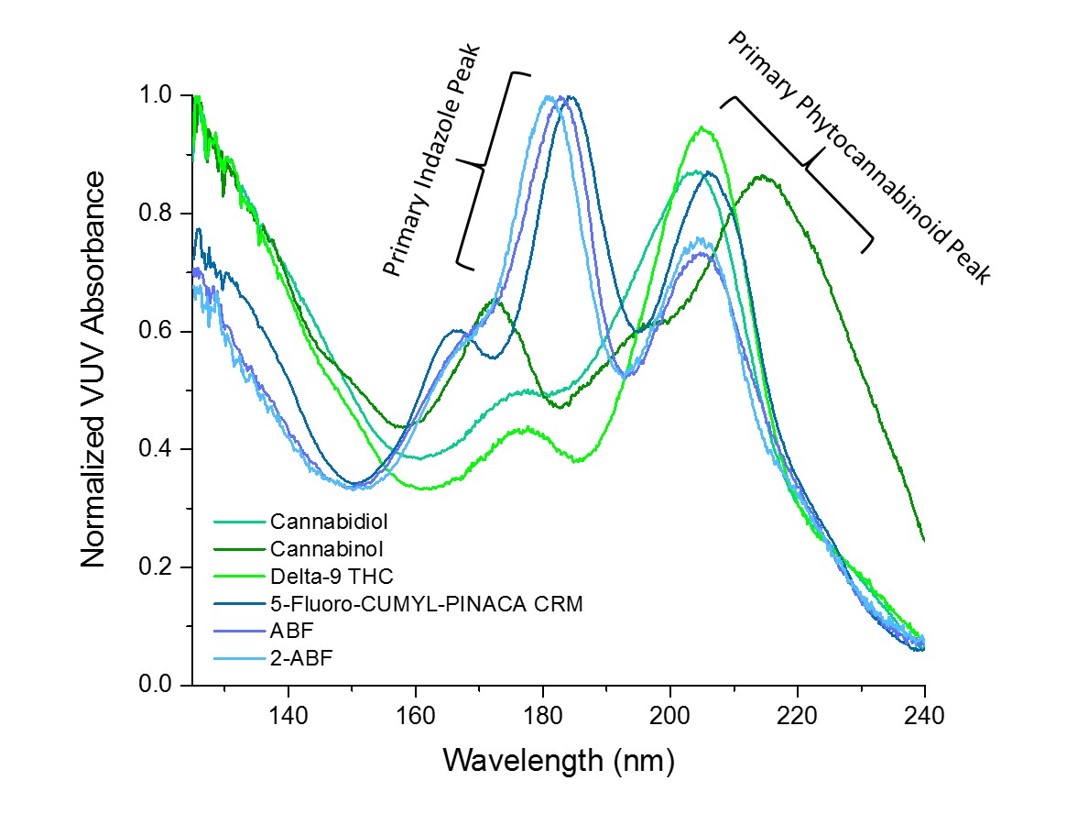
Figure 3: An overlay comparing spectral features of phytocannabinoid and indazole compound classes.
GC-VUV presents the DEA and cannabis regulators with a high throughput solution for the analysis of cannabinoids. GC separations can typically be completed in less than 15 minutes, and compound identification and quantitation through VUV software requires little time and effort. Further adding to the unique value added by VUV spectroscopy is its unmatched selectivity of isoforms and the ability to characterize compounds by their classes. It’s high time that a detector came along that was able to keep up with the rapidly changing complexities of drug enforcement and regulation.









Leave a Reply