Published Jack Cochran on July 17, 2017
I borrowed “The Lazy Analytical Chemist” part of my title from my friend and colleague Steve Lehotay, who once said in a presentation, “A good analytical chemist is a hard-working lazy person”. Steve’s point was that we chemists will work long hours just to save a few minutes in a method. For this post, I am turning his statement around a bit by declaring that I am so lazy I won’t even work on method development at all, if I don’t have to! Fortunately, I have a VUV detector (VGA-100) in my lab that allows me to get away with this sometimes, including when I’m working on residual solvent analysis in pharmaceutical products.
Recently, Lindsey Shear-Laude and I developed a static headspace – GC-VUV approach for the analysis of Class 1 and Class 2 residual solvents in 8 min. One of our industry partners reviewed the work and said, good job, but I don’t see all my solvents in there, so when are you going to do those? If we only had a non-specific flame ionization detector (FID), which is normally used for residual solvents analysis by GC, that comment might have made my head explode. That’s because the possibility of coelutions is quite high when adding analytes, which means more method development would be required. Remember my title; I’m too lazy for that.
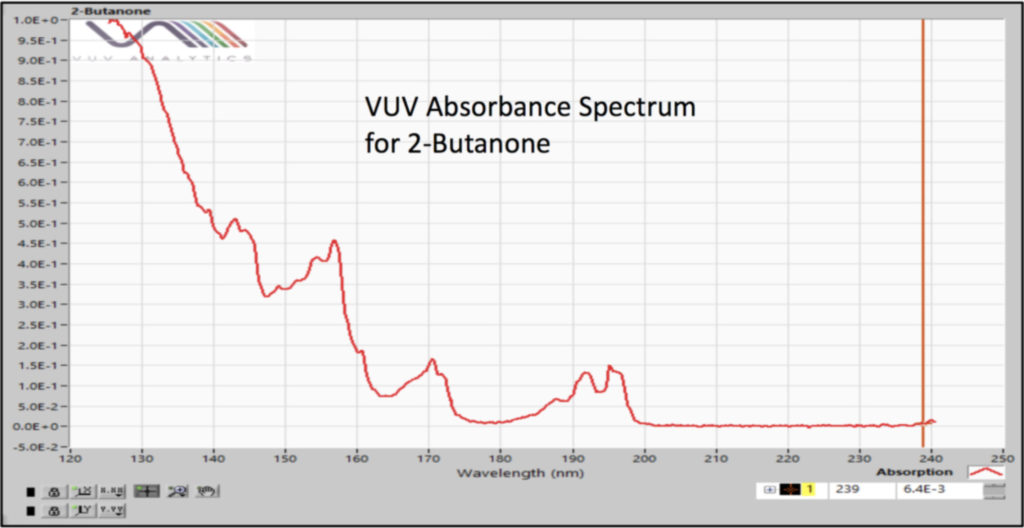
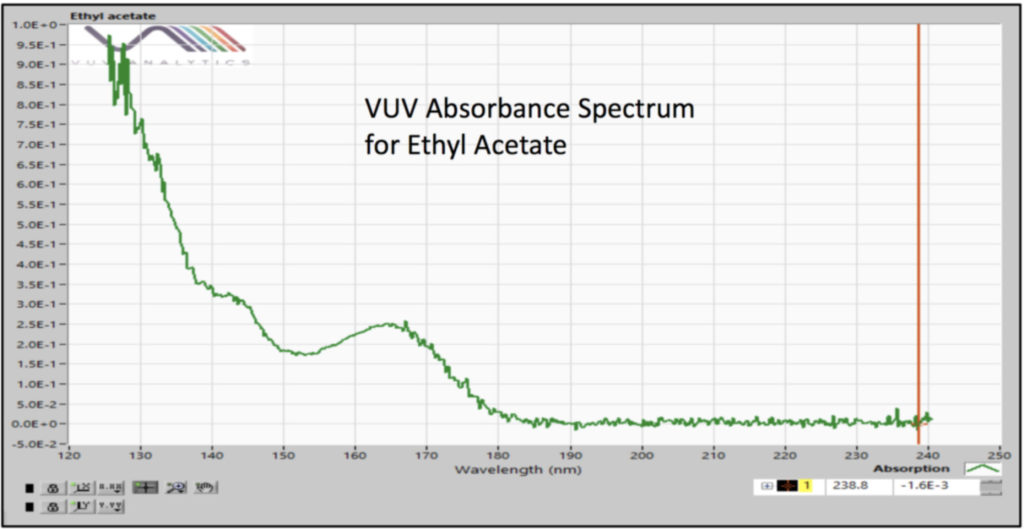
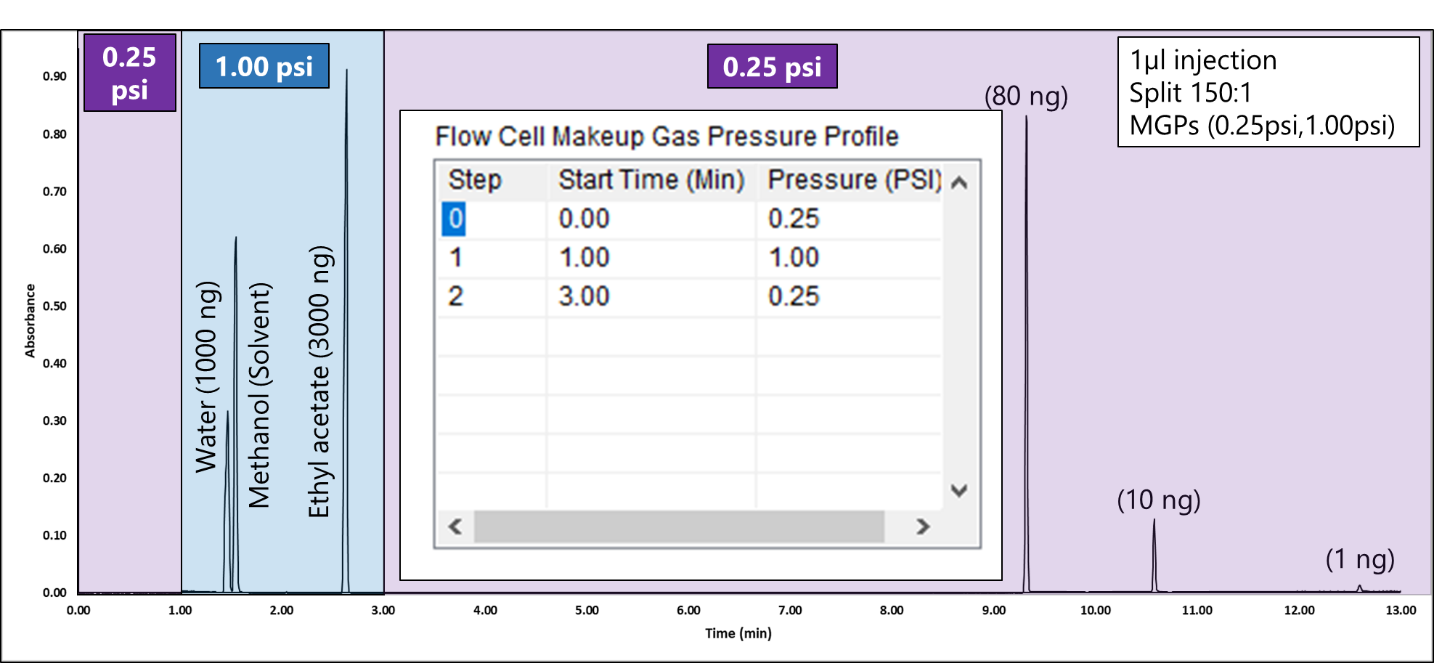
We analyzed the additional solvents, which were all Class 3, using our previously developed static headspace – GC-VUV method (Figure 1). As somewhat expected, we had a coelution, but due to the unique absorbance spectra for the two compounds of interest, 2-butanone and ethyl acetate (Figures 2 and 3), we could easily deconvolve these solvents (Figure 4) and produce excellent calibration curves spanning the 0.05x to 1x ICH concentration limits for 200 mg samples of infant’s acetaminophen in 2 mL water (Figures 5 and 6). Sloth rules!
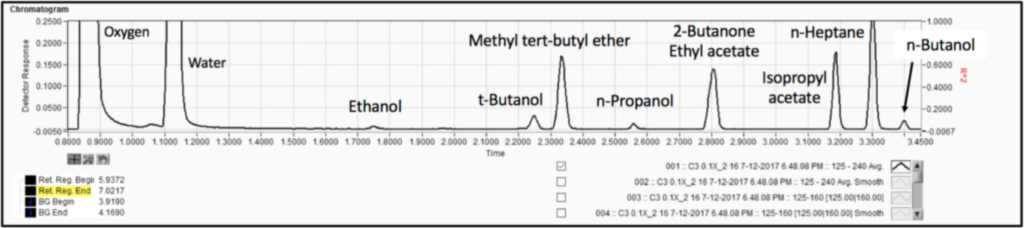
Figure 1. Static headspace – GC-VUV of Class 3 residual solvents in infants acetaminophen dissolved in water. The last compound, n-butanol, elutes in about 3.4 min.
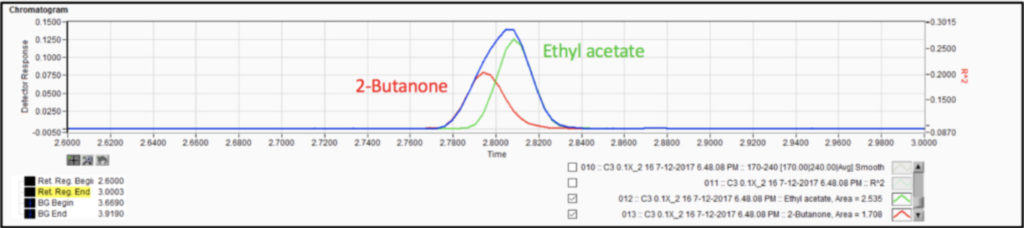
Figure 4. Spectral deconvolution of coeluting Class 3 solvents, 2-butanone and ethyl acetate, made possible by the uniqueness of their vacuum ultraviolet absorbance spectra.
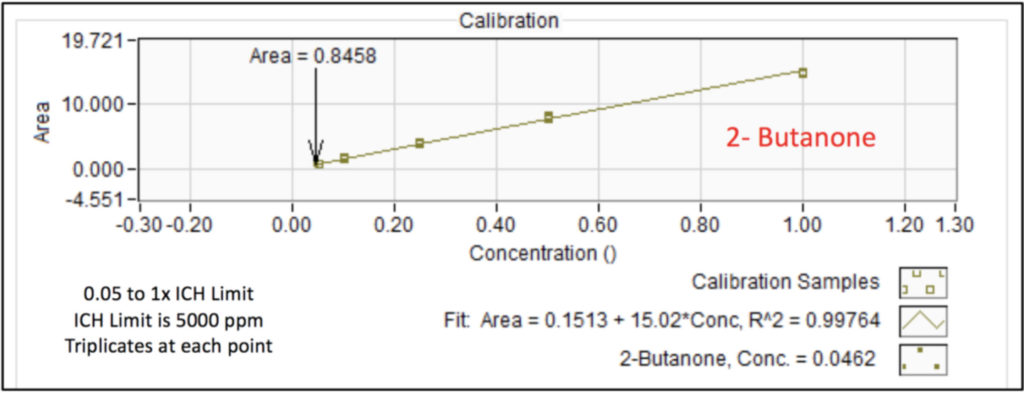
Figure 5. Calibration curve for 2-butanone in infant’s acetaminophen from 0.05 to 1x the ICH concentration limit using static headspace – GC-VUV. Triplicates were analyzed at each point.
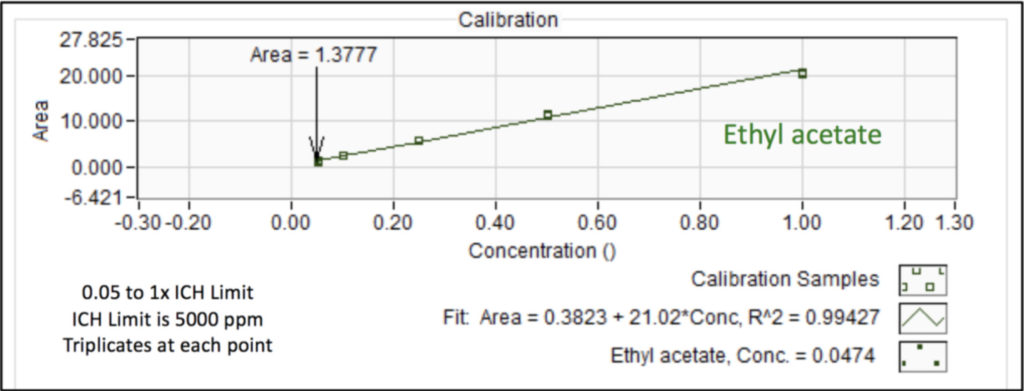
Figure 6. Calibration curve for ethyl acetate in infant’s acetaminophen from 0.05 to 1x the ICH concentration limit using static headspace – GC-VUV. Triplicates were analyzed at each point.
For more information on this work, be sure to watch the rebroadcast of Lindsey’s LCGC webinar on residual solvent analysis by GC-VUV. Register at our pharma page by clicking this link: Analysis of Residual Solvents in Pharmaceutical Excipients and Products with Static Headspace – Gas Chromatography – Vacuum Ultraviolet Absorbance Spectroscopy








Leave a Reply