Published Alex Hodgson on February 11, 2019
Part 3 – You Can’t Always Get What You Want
As mentioned at the end of Part 1, one of the more promising methods to replace UOP-326, for conjugated diolefins (CDOs) determination in fuels, comes from some recent work by the folks at ExxonMobil, which uses a similar Diels-Alder reaction but more selective dienophiles: 4-methyl-1,2,4-triazoline-3,5-dione (MTAD) and 4-phenyl-1,2,4-triazoline-3,5-dione (PTAD) (Figure 1). The derivatization reaction usually occurs within seconds at room temperature and does not have the same issues with alcohols and thiols that maleic anhydride does (though we discovered that the reaction can be incomplete for sterically hindered diolefins like 2,5-dimethyl-2,4-hexadiene). Adding these bulkier TAD molecules (113-175 Da) to lighter CDOs (68-108 Da for C5-C8) allows for good chromatographic separation from bulk gasoline-range matrix while still being GC-amenable. The method pairs well with both mass spectrometry (MS) – the adduct mass fragments are distinct for each parent diene – and nitrogen chemiluminescence detection (NCD), as the triazoline moiety gives a strong nitrogen signal. Let’s see if this derivatization method will work with GC-VUV.
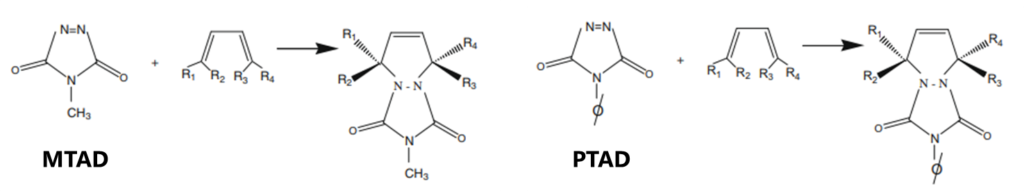
Figure 1. Schematic of the Diels-Alder reaction of MTAD and PTAD with a diene. Notice how the diene becomes a monoene in the adduct, which will change the VUV spectral profile.
In order to convert this application to GC-VUV, a couple considerations needed to be made. First, we decided to use PTAD over MTAD because we know that generally aromatics have a much better VUV absorbance than aliphatics, which could allow us to detect lower levels of PTAD adducts; we were also interested in what the PTAD adducts might look like spectrally, as PTAD contains a few heteroatoms on top of the phenyl group.
Second, a different hardware setup was required versus what we typically use for ASTM D8071, our gasoline PIONA method. On the GC side, we used a 15m Restek Rxi-5ms instead of the 30m Rxi-1ms, a higher flow rate (4 mL/min helium), and a GC oven temperature profile that ramped quickly (25°C/min) to 300°C to keep the run time relatively short (11.5 minutes) and push the bulk matrix through as quickly as possible while still getting separation for the PTAD adducts (Figure 2). For the VGA we switched to a VGA-101 because 1) the transfer line and flow cell can heat to 350°C (the VGA-100 max is 275°C, which is below our final GC oven temperature), and 2) the acquisition range extends out to 430 nm, which will catch any potential high-wavelength spectral features of these CDO-PTAD adducts. This turned out not to be the case, as the spectra for the adducts look to be mostly dominated by the phenyl group of the PTAD around 190 nm (Figure 3).
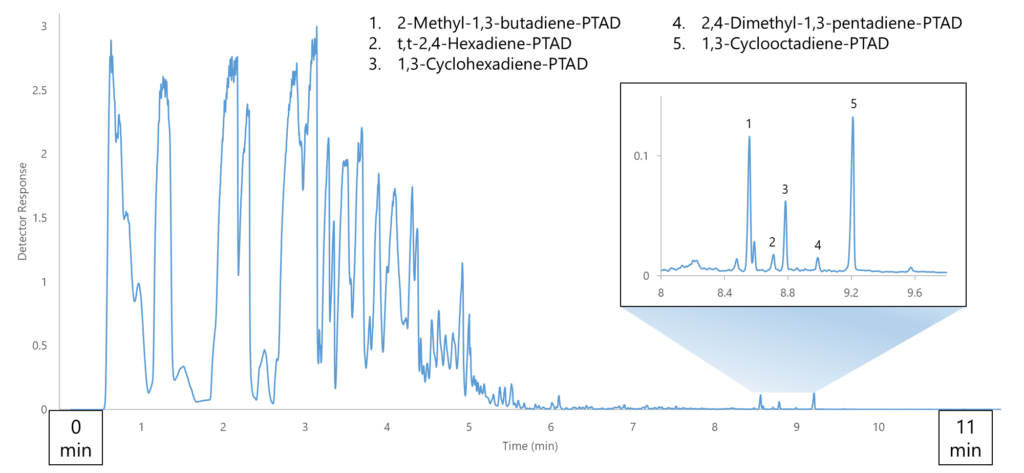
Figure 2. With this GC-VUV method, we’re pushing the bulk hydrocarbons through as quickly as possible, almost treating them as the “solvent”, while eluting the CDO-PTAD adducts afterwards. This sample was a gasoline spiked with 500 ng/μL total conjugated diolefins, derivatized with 50 μg PTAD.
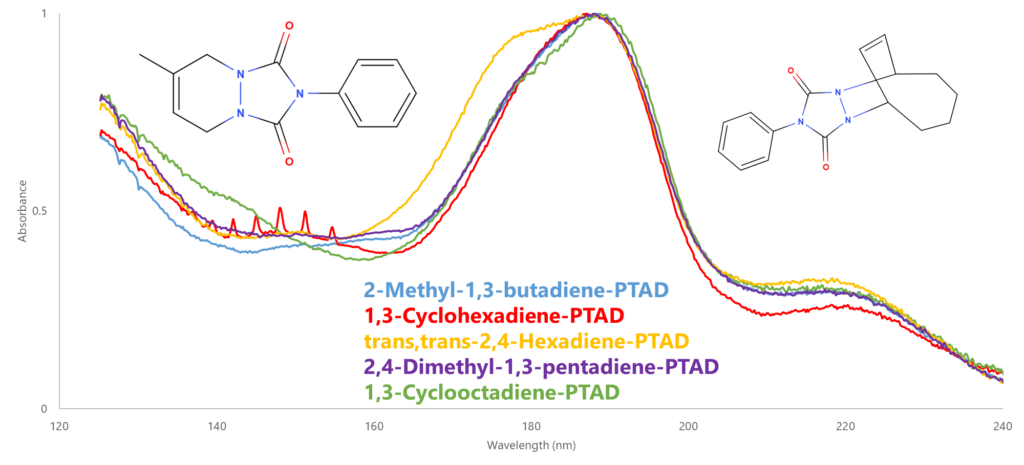
Figure 3. Looking at the molecular structure of the adducts (top left: 2-methyl-1,3-butadiene-PTAD; top right: 1,3-cyclooctadiene-PTAD), the adduct absorbance spectra are not quite as rich as anticipated past 240 nm (not shown), seemingly dominated by the phenyl group of PTAD. They still yielded a strong absorbance overall thanks to the benzene ring.
The adducts overall showed strong absorbance and, combined with them eluting a clean area of the chromatogram, allowed another order of magnitude for detection, down to as low as 10 ppm compared to 100-500 ppm from Part 2 (Figure 4). You’re probably wondering what some of those unlabeled peaks in Figure 4 are; well, so are we. One issue we encountered was knowing which CDO species would be present: if we had not done the leg work beforehand of running individual adduct standards and banking their spectra, we wouldn’t be able to identify any of the peaks. For some this may not be an issue, but it would not be conducive for uncharacterized fuels.
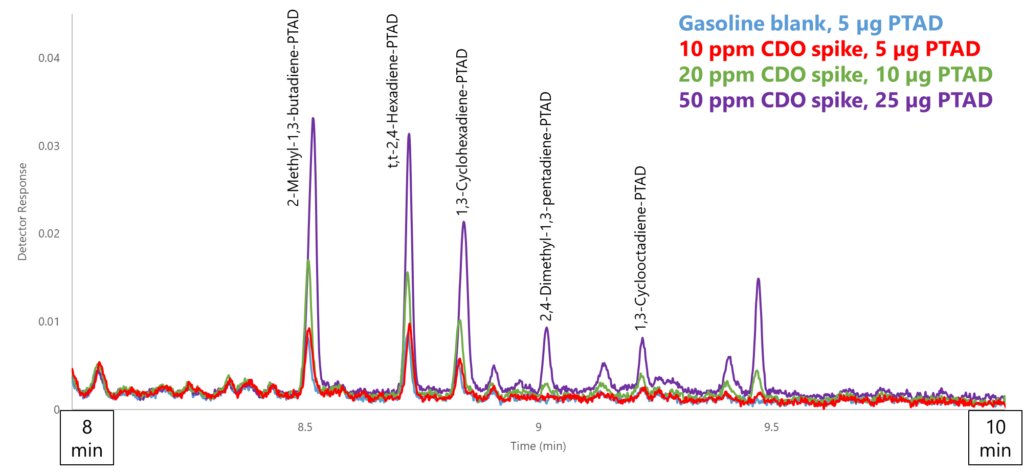
Figure 4. By adding a strongly absorbing phenyl group to each CDO and eluting the derivatized CDOs in a clean area of the chromatogram, the lower detection limit dropped from the 100-500 ppm range (ASTM D8071) to 10-20 ppm. The detection limit for the PTAD method could be lowered further with a larger injected sample volume.
The second issue is that not all the peaks are CDO-PTAD adducts. PTAD (as well as MTAD) is known to polymerize in high enough concentrations, so you need enough PTAD to fully react with all CDOs present in the sample but not so much that it reacts with itself. The self-polymerization of PTAD caused major interferences in the chromatography (Figure 5). This leads me to my next point: not only do you need to know which CDO species are present, but you need to already know the approximate total CDO concentration prior to derivatizing (unless you want to run multiple spikes at different concentrations, which seems quite inefficient to me). Again, this may not be an issue if you’re checking a well-characterized sample, but for exploratory work it would be extremely challenging.
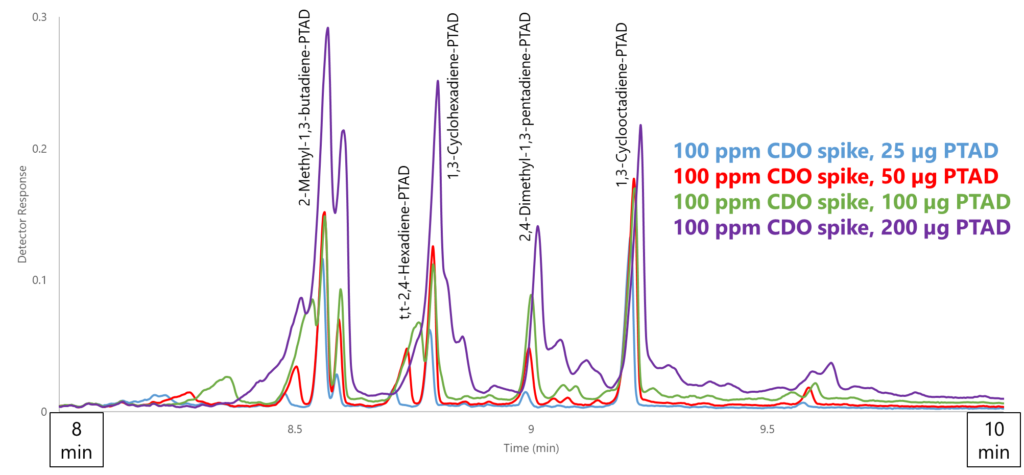
Figure 5. A big limitation of the derivatization method is the need to know the approximate concentration of conjugated diolefins prior to derivatizing, as adding too little PTAD will lead to incomplete derivatization, whereas adding too much will cause PTAD polymerization, which interferes with the chromatography. Both extremes lead to low method accuracy.
While an interesting experiment to run on GC-VUV, perhaps this derivatization method should remain an MS or NCD application. In the meantime, we’ll stick to our strength of identifying and quantitating CDOs using the tried and true D8071 method.
References









Leave a Reply