Published Jack Cochran on July 17, 2017
Gas chromatographers have been taught for ages now to pay attention to van Deemter (or at least, his equation) to get the best separations by using an appropriate linear velocity for a given carrier gas. Unfortunately, “appropriate” can be a synonym for “slow”. So, if you’re impatient like I am then you may not want to wait for all that efficiency. Fortunately, I have a handyman’s secret weapon in my lab, a vacuum ultraviolet spectrometer that can do “separation” at the end of the GC column. That allows me to compress the chromatography for higher throughput analyses, like Lindsey Shear-Laude and I recently did for residual solvents in pharmaceutical products.
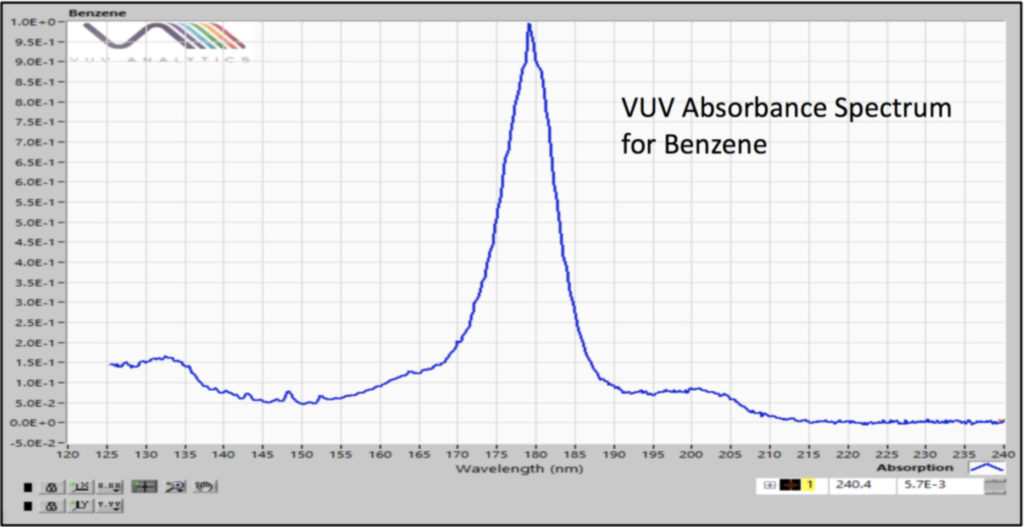
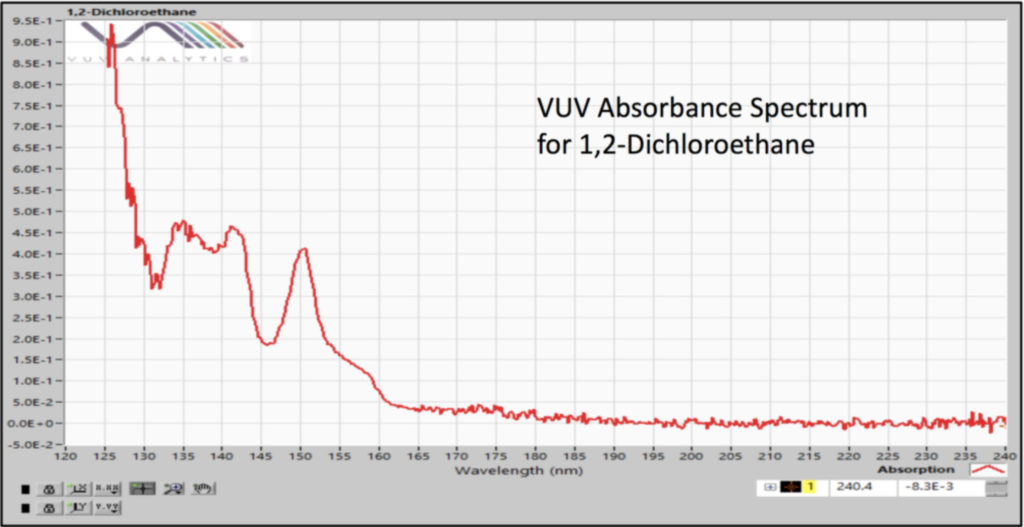
Our static headspace – GC-VUV method used a 30m x 0.25mm x 1.40µm Rxi-624Sil MS column with a constant flow of helium at 4 mL/min and a 30°C/min GC oven temperature ramp for combined analysis of Class 1 and Class 2 residual solvents. The fast carrier gas flow rate and oven temperature program allow quick elution of the compounds of interest (Figure 1) and the GC run time (8 min) matches very well with the headspace incubation time (10 min). Sure, we had coelutions, but we can deconvolve (i.e., “separate”) them (Figure 2) based on the uniqueness of their absorbance spectra (Figures 3 and 4).
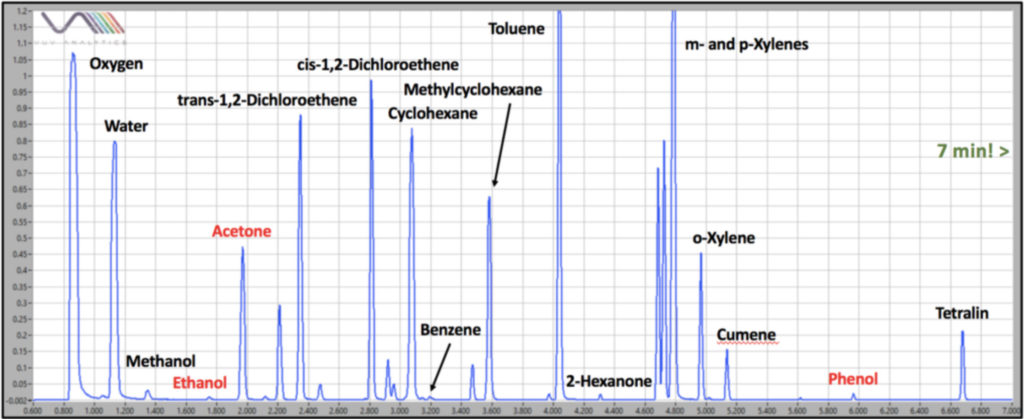
Figure 1. Static headspace – GC-VUV of 29 Class 1 and Class 2 residual solvents spiked in throat spray dissolved in water. The last compound, tetralin, elutes in about 6.7 min. The ultra-fast analysis is afforded by a high helium carrier flow rate of 4 mL/min, a fast oven temperature ramp of 30°C/min, and the ability to deconvolve coeluting analytes with vacuum ultraviolet spectroscopy. Not all solvents are labeled on the chromatogram. Those in red are non-target compounds in the throat spray.
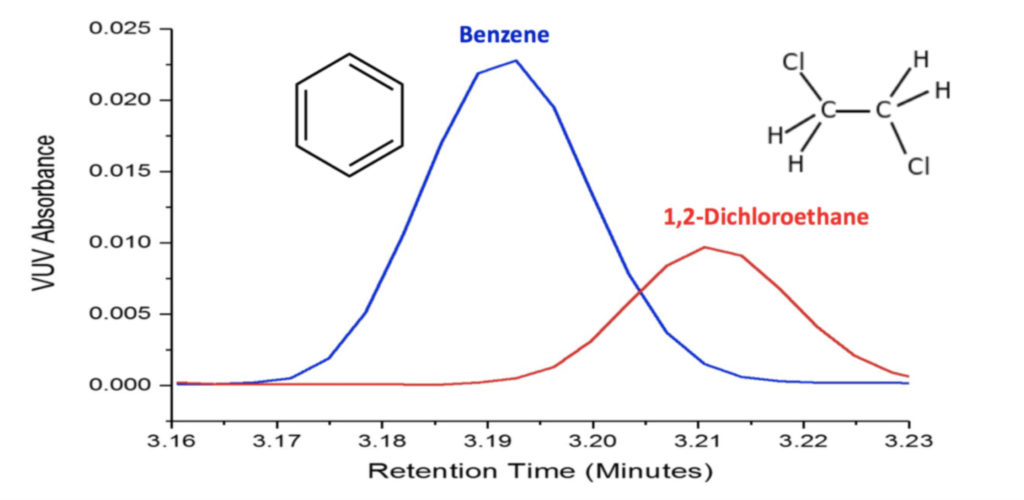
Figure 2. Vacuum ultraviolet absorbance spectral deconvolution of coeluting Class 1 residual solvents, benzene and 1,2 dichloroethane, gives pure peaks for accurate quantification.
If you want more details on this work than I can give in a short blog post, be sure to tune into the rebroadcast of Lindsey’s LCGC webinar. Visit our pharma page to view the webinar and follow-up presentation with GERSTEL, INC. https://vuvanalytics.com/industry/pharma/
Other reading on residual solvent analysis using static headspace – GC-VUV:
GC-VUV of Residual Solvents – The Lazy Analytical Chemist
GC-VUV of Residual Solvents – No Matrix Effects with the Gerstel MPS2 Static Headspace Autosampler
GC-VUV of Residual Solvents – Non-Target Analysis
GC-VUV of Residual Solvents – Improving Static Headspace Sampling with Water
GC-VUV of Residual Solvents – Matching a Fast Autosampler with a Fast Absorbance Spectrometer
Rapid Equilibration of Samples for Residual Solvents Analysis by Static Headspace – GC-VUV







Leave a Reply